Summary
- MYOK may have seemed an expensive and unwanted use of cash at first, but it couldn't have been too bad for management to have bought it.
- Indeed, successfully raising clinical awareness for HCM and R&D successes will have made the acquisition a good bargain.
- We can determine this with a market assessment based on BMY pricing power with Eliquis, which manages similar patient risks.
- Overall, the market has determined the MYOK price fair, and we see substantial upside from the acquisition if BMY executes.
The $13.1 billion of MyoKardia (NASDAQ:MYOK) left some investors in Bristol Myers Squibb (NYSE:BMY) skeptical. Indeed, the company had not long ago served up heaps of cash to buy Celgene, seen as a promise of the end of inorganic growth with the pipeline finally being shored up. In addition to the MyoKardia acquisition being perceived as a breaking of this promise, it did not seem a great bargain either based on the rather limited number of diagnosed patients. However, on a deeper look, the acquisition seems rather promising. Using Eliquis pricing, it seems that, with strong execution, the acquisition would have been a bargain, and the biotechnology could have wider applications. Overall, the acquisition seemed appropriate, with management's decision to continue inorganic growth merely opportunistic in this case rather than strategically imperative. We continue to look favourably on BMY as a buy.
Mavacamten and HCM
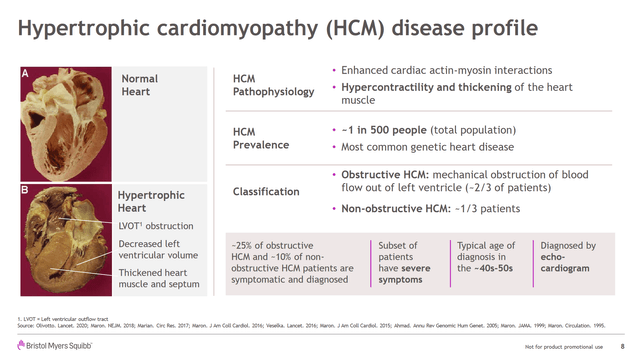
Hypertrophic cardiomyopathy (HCM) is a genetic heart condition whereby the walls of the ventricle are overly thick, leading to a tendency for hyper-contractility which can lead to heart failure and sudden death. Mavacamten, which is in Phase III, is MyoKardia's drug which acts on heart muscle cells by getting in the way of the actin-myosin cross-bridge to reduce the intensity of heart contractions. For now, the drug's indication is for obstructive HCM.
(Source: MYOK Acquisition Presentation)
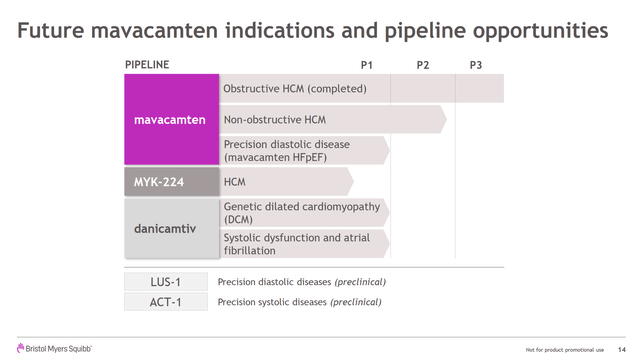
There is also non-obstructive HCM as an indication for which is in Phase II and the possibility of using the precise biotechnology for other diastolic and systolic diseases. Indeed, they are at the pre-clinical stage with some of these indications, and in Phase I with others.
(Source: MYOK Acquisition Presentation)